Personal Injury
16 Questions to Ask a Personal Injury Lawyer
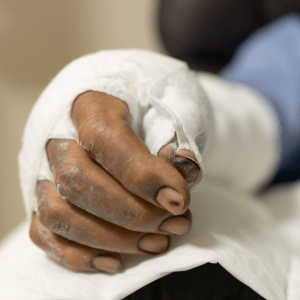
It can be quite difficult to secure compensation for losses such as injuries after serious accidents. You might have to...
Workers’ Comp vs. Personal Injury Claims
If you've been injured, particularly at work, you may be wondering about workers' comp vs. personal injury. You may be...
All About Passenger On Passenger Assaults On Buses
Passengers being attacked on buses is not a new phenomenon. However, these attacks are, unfortunately, becoming more common. California has...
Blog,Personal Injury,Slip & Fall
Personal Injury Attorney Weighs In on Home Alone’s Scariest Injuries
It's the most wonderful time of the year. It's the holidays. Unfortunately, the hustle and bustle of the holiday season...
Lawsuit Seeks To Hold NFL, ESPN, Others Liable For Football Player’s Injury
Aaron Patrick, a linebacker for the Denver Broncos, filed a lawsuit on November 15 against the NFL, ESPN, the Los...
Auto Accidents,Blog,Car Accidents,Personal Injury
Diffuse Axonal Injury After a California Car Accident
A diffuse axonal injury (DAI) is one of the most severe brain injuries a person can sustain and may result...
Christmas Tree Fire Risk and Safety Tips
A Christmas tree is an important part of Christmas celebrations in many homes, workplaces and other commercial settings. It symbolizes...
The Dangers of Deep Frying Turkeys: Everything You Need to Know
Thanksgiving is full of delicious eats, but the centerpiece of the occasion is typically a large juicy turkey cooked to...
Concussion Lawsuit Against NCAA Could Be First To Go To Trial Before A Jury
The concussion lawsuit filed by the widow of former University of Southern California (USC) linebacker Matthew Gee against the National...
How Law Firms Are Embracing Technology
Technology in the legal field has come far in the past few decades, but not all law firms have kept...
- 1
- 2
- 3
- 4
- Next Page »